


Welcome!
The Materials and Systems Engineering Laboratory (MaSEL) at the University of Illinois at Chicago is led by Prof. Meenesh R. Singh. We are developing state-of-the-art materials, computational and experimental tools to solve problems related to energy, environment and healthcare. Our current research projects are steered towards solving grand challenges of the 21st century-
i) Develop Carbon Sequestration Methods
ii) Manage the Nitrogen Cycle
iii) Provide Access to Clean Water
iv) Engineer Better Medicines
Latest News & Announcements
(02/26) Congratulations to Amey Thorat, Rohan Sartape, Rohit Chauhan and Rashmi Mishra for their publication
(02/26) Congratulations to Prem K. Reddy, Anish V. Dighe, and Rajan R. Bhawnani for their publication.
(02/26) Earlier this year, Dr. Rohan Sartape successfully defended his doctoral thesis, marking the culmination of an outstanding PhD journey. His groundbreaking research focused on carbon capture and CO2 conversion, offering vital solutions for a greener future. Dr. Sartape’s work represents a significant stride in the field, leaving a lasting impact on sustainable engineering.
(02/26) Congratulations to Rashmi Mishra, Rohan Sartape and Rohit Chauhan for their publication.
(02/26) Congratulations to Rohit Chauhan, Rohan Sartape, Rashmi Mishra, Amey Thorat for their publication.
"Trends and Limits of CO2 Capture Capacities of Liquid and Solid Sorbents at Ambient Conditions," is now published in Journal of CO2 Utilization.
(02/26) Congratulations to Rohan Sartape, Rohit Chauhan, Samdavid Swaminathan, Ishita Goyal, Amey Thorat, Jindal K. Shah for their publication.
Research Highlights

Salting-Out Artificial Photosynthesis
"Chemical engineers from the US have put forward a concept for a new type of artificial photosynthetic system to convert carbon dioxide into almost pure liquid ethanol fuel. It uses a saturated salt electrolyte, and, according to their calculations, the system would be capable of generating 15.27 million gallons of ethanol per year per square kilometre."

Artificial Photosynthetic System for High-Yield Production of Ethanol
"A team at the Joint Center for Artificial Photosynthesis (JCAP) at Lawrence Berkeley National Laboratory and UC Berkeley is proposing an artificial photosynthesis scheme for direct synthesis and separation to almost pure ethanol with minimum product crossover using saturated salt electrolytes."

Moving Artificial Leaves Out of the Lab and Into the Air
"..researchers from the University of Illinois at Chicago have proposed a design solution that could bring artificial leaves out of the lab and into the environment. Their improved leaf, which would use carbon dioxide — a potent greenhouse gas — from the air, would be at least 10 times more efficient than natural leaves at converting carbon dioxide to fuel."
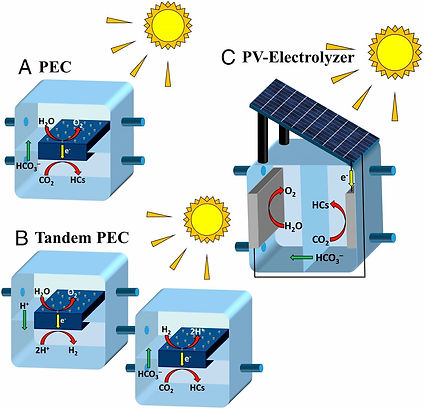
This is How Good Solar-to-Fuel Conversion Can be
"Researchers with the Joint Center for Artificial Photosynthesis at Lawrence Berkeley National Laboratory report today in the Proceedings of the National Academy of Sciences that they have evaluated the potential efficiencies of this process for several different photoelectric cell configurations, catalysts, and fuel end products. The group concluded that solar energy could break down CO2 into synthesis gas—a blend of hydrogen and carbon monoxide that used to make other hydrocarbons—at an efficiency of 18.3 percent or could make liquid hythane—a mix of hydrogen and methane—at 20.3 percent efficiency."

Converting Carbon Dioxide to Carbon Monoxide using Water, Electricity
"Researchers at the University of Illinois at Chicago and the Joint Center for Artificial Photosynthesis have determined how electrocatalysts can convert carbon dioxide to carbon monoxide using water and electricity. The discovery can lead to the development of efficient electrocatalysts for large-scale production of synthesis gas -- a mixture of carbon monoxide and hydrogen."

Breach of 'dancing' barrier governs crystal growth
"Researchers found that when crystal-forming molecules are surrounded by a solvent, like water, the solvent molecules form a shield that they call a solvation shell. When this shield fluctuates, molecules can break free to form crystals. They also showed that temperature, solvent type and the number of solvent molecules all affect the shell’s fluctuation..."

Producing Ammonia with Smaller Carbon Footprint
"Singh and colleagues have developed a new method to produce ammonia that relies on the use of a mesh screen coated in copper – a catalyst that helps bind nitrogen to hydrogen to make ammonia. The electrification of the screen helps drive the reactions at ambient conditions."




